 |
|
 |
| Mission | Search | Acknowledgements | Discussion Group | Contact Us | Links | ||
| ||||||
|
Maine DEP Monthly
Maine and WEF's
Penobscot Watershed and Development of a TMDL
Maine Rural Water
Maine
Wastewater Control Association |
AERATED
LAGOON TECHNOLOGY by Technical Note Number 8 FACULTATIVE LAGOONS:
|
|
1.) Over a time frame
of 24 hr or longer, shallow facultative lagoons can be considered as mixed
systems. |
|
2.) Facultative
lagoons are primarily anaerobic systems with an aerobic top layer that all
but disappears for at least a few hours during the night. |
|
3.) The fact that
dissolved oxygen virtually disappears after photosynthesis ceases indicates
that the surface oxygenation rates are low compared with the oxygen demand
in the water column. |
|
4.)The fact that a high pH is sustained during photosynthesis indicates that the rate of carbon dioxide transport from the atmosphere is low compared to the rate at which algae are utilizing the carbon dioxide. |
Distinct seasonal variations occur. During the winter months the diurnal changes in the lagoon were minimal compared to those observed during the summer.
NUISANCE BEHAVIOR
Facultative lagoons are often the source of odors. The odors are primarily
caused by two factors - mats of dead algae decomposing at the surface and along
the sides of the lagoon, and hydrogen sulfide evolving from the lagoon.
Decaying algal mats give rise to a pig pen odor. Such mats are caused by
the periodic occurrence of excessive quantities of filamentous, blue-green
algae. This group of algae which flourishes in facultative lagoons during the
summer months does not settle like green algae but floats and accumulates at the
surface where it decays in the sun, giving off noxious odors. As stressed
ecosystems, facultative lagoons are subject to periodic algal blooms.
Hydrogen sulfide is formed from sulfates in wastewaters that become reduced in the anaerobic environment in the lagoon, as well as from the decomposition of proteinaceous materials in the bottom solids. Hydrogen sulfide, which has a rotten egg odor, ionizes in solution to form the equilibrium
H2S " H+ + HS- (1)
In as much as hydrogen ions are one of the ionic species involved in the equilibrium, the particular form in which the sulfide exists is dependent upon the pH of the system. This dependence is illustrated in Fig. 3. The combined form, H2S, is the only form of the sulfide that is not ionic and can be released from solution as a gas. From the figure it is noted that H2S exists in significant concentrations only at pH values less than 8. Since the top layer of a facultative lagoon during daylight hours in the summer is greater than 8, the layer functions as a lid to retain the sulfide in solution. Only during the night hours, when the pH of the top layer falls below 8 can hydrogen sulfide escape from the lagoon.
DISCUSSION
Facultative lagoons have three major disadvantages: odors, variable
effluent quality, and large land-area requirement. The sources of odors have
been discussed above. Variable effluent quality is a characteristic of all
facultative lagoons. The removal of BOD5 will vary from 50 to 95 percent,
depending on how much algae is in the lagoon at the time. Only with additional
treatment such as intermittent sand filtration can facultative lagoons
consistently achieve BOD5 effluent limits now being imposed. Furthermore
significant concentrations of nitrogen and phosphorus are found in the
effluents. As for land area requirements, a wastewater flow of 1 mgd will
require about 30 acres of lagoon, if the latter is loaded at 50 lb
BOD5/acre-day.
Consideration of the characteristics of facultative lagoons and their performance provide insight to the behavior of aerated lagoons that do not have the aeration, mixing, size, or depth that they should have. Such insight will provide a guide for design modifications that should be made to the aerated lagoon to achieve optimal performance.
REFERENCE
Meenaghan, G. F. and Alley, F. C. (1963). "Evaluation of waste stabilization pond performance." 3rd Annual Industrial Water and Waste Conference, Rice University, 3-9.
(END OF NOTE)
Fig. 1. Conversion of organic-C to biomass in facultative lagoons.
Fig. 2. Profiles typical of summer conditions.
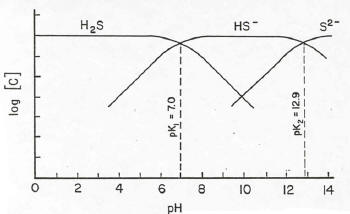
Fig. 3 Hydrogen sulfide system.
|
|
Effluent BOD5 - A Misleading Parameter For the Performance of Aerated Lagoons Treating Municipal Waste |
|
|
Aerated Lagoon Effluents |
|
|
Control of Algae |
|
|
Nitrites and Their Impact on Effluent Chlorination |
|
|
Aerated Lagoons for Secondary Effluent |
|
|
Nitrification in Aerated Lagoons With Intermittent Sand Filters |
|
|
Mixed Liquor Recycle (MLR) Lagoon Nitrification System |
|
|
Facultative Lagoons - A Different Technology |
|
|
Sludge Accumulation in High Performance Aerated Lagoon Systems |
|
|
Ammonia Feed Back in the Sludge of a CFID Nitirification System |
|